Baby Size and Fetal Movement at 37 Weeks
- Research article
- Open Access
- Published:
Correlation study betwixt increased fetal movement during the third trimester and neonatal outcome
BMC Pregnancy and Childbirth volume xix, Article number:467 (2019) Cite this article
Abstract
Groundwork
We aimed to analyze the correlation between increased fetal movements in the tertiary trimester and neonatal outcomes.
Methods
We enrolled pregnant women (n = 219) who reported increased/excessive fetal movements in the third trimester in our hospital. A command group of salubrious women (n = 278) who had undergone regular childbirth and commitment in our infirmary during the same menses and did not report abnormal fetal movements were besides recruited. All pregnant women underwent fetal non-stress exam. Nosotros analyzed the neonatal weight, advent, pulse, grimace, activeness, and respiration score, degrees of amniotic fluid contamination, amniotic fluid volume, conditions of umbilical cord around the neck and cord length, and incidence of modest for gestational historic period. In addition, the incidence of preterm delivery, cesarean section rate, postpartum hemorrhage, and other postpartum complications were also analyzed. Nosotros then analyzed the correlation betwixt increased/excessive fetal activity and neonatal outcomes.
Results
Women with complaints of increased/excessive fetal movements exhibited increased fetal movements mainly around 31 and 39 weeks of gestation. Several pregnancy variables, including number of previous commitment, gestational historic period (less than 34 weeks and more than 37 weeks) and vaginal birth rate, were associated with increased/excessive fetal movements. In improver, women who reported increased/excessive fetal movements had higher odds of big for gestational age (LGA), especially those with gestational historic period over 37 weeks.
Conclusion
Increased/excessive fetal movements may be used to predict adverse neonatal upshot such every bit LGA.
Background
Globally, 2.6 one thousand thousand infants were stillborn each year [ane]. Hence, many efforts have been invested to report modifiable risk factors, which could be used every bit targets to introduce intervention to alleviate the risk of stillbirths. These efforts were mainly retrospective studies with example-controls to investigate risk factors such as fetal movements due to mother'southward experience, sleep position during maternity, maternal intuition, exercise, and diet [2,3,4]. These studies have confirmed that fetal abnormalities can be detected early by monitoring fetal movement, which are a common complaint by significant women during the 3rd trimester. Furthermore, these studies reported that reduced fetal motion is a clear maternal symptom associated with stillbirth and poor prognosis of neonates [5,6,7,8], possibly through placental dysfunction [9].
While reduced fetal movements often crusade concern and feet among women and also required clinical assessments [6], clinical data regarding the human relationship of increased/excessive fetal movements and prognosis of neonatal outcomes are currently non adequately bachelor. A questionnaire study in Nigeria establish that women tend to limited concern on excessive movement in a significantly higher rate than on reduced movements (31.1% vs. 21.8%) [ten]. Hence, it is important to study whether excessive fetal movements are associated with whatsoever adverse neonatal outcomes. This information would be used to guide medical professionals in providing nursing to women, and would also allow more detailed information to be provided to women in order to reduce maternal feet.
To this end, we analyzed the neonatal weight, appearance, pulse, grimace, activity, and respiration (APGAR) score, degrees of amniotic fluid contamination, amniotic fluid volume, conditions of umbilical string around the neck and cord length, and incidence of small for gestational historic period of meaning women, with or without complaints of increased fetal movements. In add-on, the incidence of preterm delivery, cesarean department rate, postpartum hemorrhage, and other postpartum complications were also analyzed. We so reported the correlation betwixt increased/excessive fetal activity and neonatal outcomes in this study.
Methods
Study participants
A cohort study was conducted by prospectively recruiting women who were admitted to the Emergency Department of Obstetrics, Shanghai Sixth People's Hospital, Shanghai Jiao Tong University with a complaint of increased/excessive fetal movements from April 1st 2017 to November 30th 2017. Our Emergency Department provides 24-h service and has the capability to assess approximately 3000 women per yr. Nosotros enrolled pregnant women (n = 219) who reported having increased/excessive fetal movements in the third trimester in our hospital. We besides included a control group of healthy women (n = 278) who had undergone regular childbirth and delivery in our infirmary during the same period and did not report abnormal fetal movements. All significant women underwent fetal not-stress test (NST).
Inclusion criteria were: 1) women over 28 weeks' gestation; 2) with delivery record in our hospital during the study period. Exclusion criteria were: ane) women who had antenatally-diagnosed congenital anomalies and/or had multiple pregnancy; 2) with medical weather condition requiring medication that could bear upon fetal movements during the report.
Fetal condition was measure out by attention staff using NST at our Obstetrics Emergency Department. After delivery, chart review was performed past the authors, and the pregnancy characteristics and outcomes were evaluated. Demographics data (e.g. marital status and maternal historic period) and characteristics related to pregnancy (e.g. number of previous deliveries, gestational historic period, maternal weight at concluding calendar week of gestation) were collected, and several run a risk factors for fetal prognosis outcomes (e.thousand. hypertension more than 140/xc mmHg, and diabetes) were assessed. Finally, delivery (e.thousand. cesarean department charge per unit, neonatal weight, APGAR score, degrees of amniotic fluid contamination, amniotic fluid volume, conditions of umbilical cord around the neck and string length, and incidence of pocket-size for gestational age) and postpartum complications (e.1000. postpartum hemorrhage, Neonatal Intensive Care Unit of measurement admission) information were likewise obtained. The APGAR score was determined by evaluating the newborn baby on five simple criteria on a calibration from nix to two, then summing up the five values. The degrees of stained amniotic fluid were divers as follows:
- 1.
Class i: a pocket-size amount of meconium in a large corporeality of amniotic fluid, with slightly green or yellowish discoloration.
- two.
Grade two: a moderate amount of meconium in a off-white amount of amniotic fluid, with clear meconium staining shown as 'khaki green' or brownish in color.
- 3.
Grade three: a large amount of meconium in a reduced amount of amniotic fluid, with heavy meconium staining shown as thick with 'pea soup' consistency.
Information assay
Women with increased/excessive fetal movements were compared to the control group of women. Predictive Analytics Software eighteen (IBM, 2009) was used for data analysis. Continuous data were analyzed using the t-exam. Chiselled information were analyzed using Pearson'due south Chi-squared examination. Multivariable logistic regression was and so used to model the association between increased/excessive fetal move and neonatal weight and pre-term delivery. In the model, we controlled for variables and covariates known to be associated with neonatal weight and pre-term delivery, including gestational age, maternal weight at last week of gestation, delivery mode, and hypertension.
Results
From April 1st 2017 to Nov 30th 2017, in that location were a total of 219 women recruited via the Obstetrics Emergency Department in our infirmary with a complaint of increased/excessive fetal movements. We as well included a total of 278 healthy women who had undergone regular childbirth and delivery in the Department of Obstetrics at our infirmary during the same menstruum and did not written report aberrant fetal movements. Equally a event, we analyzed a full of 497 women who had commitment in our hospital. In full general, virtually 9.6% of pregnant women who were treated in our hospital reported experience of increased/excessive fetal movements. The characteristics of the analytic accomplice are shown in Table one. Subjects were largely nulliparous (67.viii%). We also found that hypertensive affliction of pregnancy (12%) and diabetes (8%) were the most common comorbid weather condition. The rates of pre-term, small-for-gestational-historic period neonates, postpartum hemorrhage (more than 500 ml), Neonatal Intensive Care Unit admission were relatively low. In women with a complaint of increased/excessive fetal movements, increased fetal movements appeared mainly betwixt 31 and 39 weeks of gestation (Fig. ane).
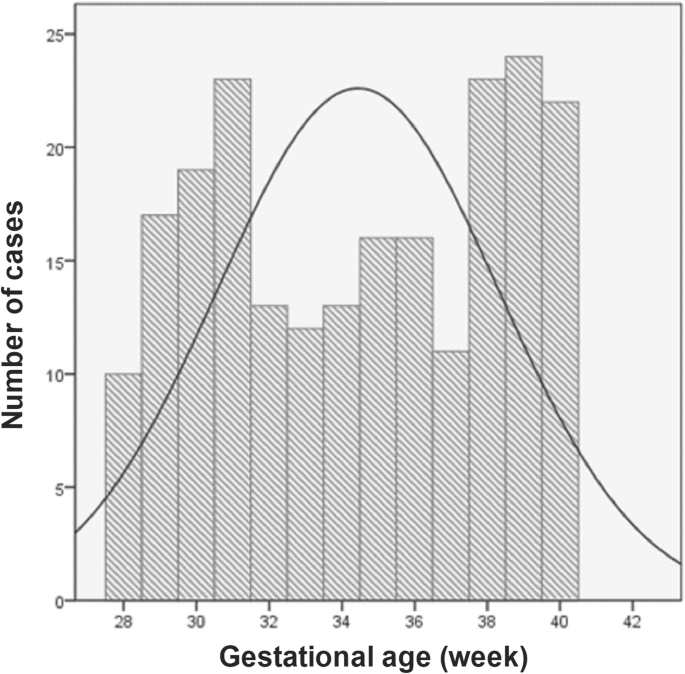
Number of women who experienced increased/excessive fetal movements during the third trimester of pregnancy
Using chi-foursquare tests, we found that several pregnancy variables were associated with increased/excessive fetal movements (Table ii): number of previous delivery [odds ratio (OR) 2.2, 95% confidence interval (CI) one.v–3.0], gestational historic period (less than 34 weeks and more than 37 weeks; OR 2.1, 95% CI 1.4–three.ane) and vaginal birth (OR 1.seven, 95% CI 1.ane–two.nine). In improver, we found that women who experienced increased/excessive fetal movements had less incidence of pre-term delivery (OR 1.4, 95% CI 1.0–two.1), and the newborns had higher neonatal weight (OR 2.five, 95% CI i.half dozen–3.2) (Table 2).
In the multivariable model estimating the association between increased/excessive fetal movement and neonatal outcomes (Table iii), there were higher odds of big for gestational age (LGA) in women who had increased/excessive fetal movements (adjusted OR 1.86, 95% CI 1.69–2.07). Higher odds of LGA were also associated with women who had increased/excessive fetal movements and gestational age more 37 weeks (adapted OR 1.98, 95% CI 1.35–2.24).
Discussion
In the present study, we found that the percentage of women who reported increased/excessive fetal movements was relatively higher in those who had the first-time pregnancy compared the command group. This incidence rates dropped in those who had a history of pregnancy. This phenomenon may be due to the first pregnancy, during which pregnant women are not experienced in the method of counting fetal move and appear more sensitive and cautious during counting. Hence, these first-fourth dimension pregnant women may report increased fetal movements more ofttimes, which might compromise the objectivity of data in the current report. In fact, increased/excessive fetal movement is quite a common feel later on 37 weeks of gestation. Therefore futurity investigations should include more objective measurements to address this limitation.
I important finding in our study is that, near of the neonates with increased fetal movement did non show poor prognosis. Instead, we observed better prognosis, which was reflected in the lower percentage of pre-term infants in women who had increased fetal movement. The neonatal weight of the newborns was significantly higher in women who had increased fetal movement than those of the control group. Yet, this may exist too due to a college percentage of pre-term infants in the control grouping, resulting in a decrease in the weight of newborns in the control group. Furthermore, in women who had increased fetal movements, nosotros observed that increased fetal movements appeared mainly betwixt 31 and 39 weeks of gestation. This increased fetal move at 31 weeks of gestation is likely considering that fetal weight gain is in an accelerated period, when the fetal movement may exist frequent and obvious. At the 39th week of pregnancy, the frequency of imitation contractions increased, coupled with the proximity of childbirth. These factors may also contribute to the enhanced consciousness of fetal movement in women. Furthermore, our findings that well-nigh 9.six% pregnant women reported experience of increased/excessive fetal movements were similar with previous reports. In the state of affairs, task, actions, and results (STARS) cohort study, 1714 women were recruited from more seven countries, and eight.5% of respondents reported excessive fetal movements [11]. Among the respondents, the symptom frequency from the 4 major countries who participated in the survey remained consequent. Similarly, the frequency of perceived excessive fetal movements was close to that of the 10% of women who was analyzed in a stillbirth study in Sweden [12]. Interestingly, similar to our report, the perception of excessive fetal movements was reported more frequently (12% of respondents) when gestation was beyond 37 weeks [12]. Together with our reports, these results suggested that women in the 3rd trimester of pregnancy may encounter more frequent excessive fetal movements.
Another of import finding in our written report is that, our regression analysis establish that higher odds of LGA were associated with women who had increased/excessive fetal movements and gestational historic period over 37 weeks. I of the master adventure factors of LGA is poorly-controlled diabetes, particularly gestational diabetes, also as pre-existing diabetes mellitus [thirteen,14,xv,16], which increase maternal plasma glucose and insulin levels, as well as stimulates fetal growth. In addition, studies accept shown that gestational historic period more than 40 weeks and excessive maternal weight gain can increment incidence of LGA [xv]. Consistent to this literature, our study confirmed that gestational age over 37 weeks was associated with increased incidence of LGA. Furthermore, women with increased/excessive fetal movements also had college incidence of LGA, particularly when the gestational age was more than than 37 weeks.
Changes in amniotic fluid may betoken fetal intrauterine hypoxia, fetal acidosis in the fetus, and the abnormal fetal growth [17, 18]. Surprisingly, nosotros did not observe any departure in amniotic fluid book or degrees of stained amniotic fluid in women who had increased fetal movement when compared to those of the control group. In add-on, there was no correlation between increased fetal activeness and the incidence of umbilical string around the neck. These results suggested that amniotic fluid book and contagion and the incidence of umbilical cord around the cervix may non be the contributing factors in increased fetal movements.
While our written report did not report increased cases of stillbirth in women who experienced increased/excessive fetal movements. Case-control studies have been performed in society to examine if there is whatsoever difference in the frequency of increased/excessive fetal movements between pregnancies with live births and those with stillbirth. In the Auckland Stillbirth Study, women who had stillbirth experienced more unmarried episodes of excessive fetal activity [19]. However, more than one episode of vigorous fetal activity was less likely perceived past those who had stillbirth. In addition, women who had stillbirth were less probable to study the perception of increased fetal movements compared to controls [19]. Furthermore, one episode of vigorous action was more likely perceived by women who had stillbirth in the STARS written report [20]. Hence, these results suggest that a sudden episode of excessive fetal activity may indicate the compromise of fetus that is related to the disturbance of environment in uterus. Future studies with a larger accomplice will be necessary to examine this hypothesis.
Decision
Our study is the first prospective study to investigate the correlation betwixt incidence of excessive fetal activity and neonatal outcomes. Historically, the doubt regarding measuring and reporting excessive fetal movements has led to focusing on reduced fetal movement and its effects on perinatal mortality and morbidity [21, 22]. While our written report found no evidence for the clan of excessive fetal movement with perinatal mortality, we did report adverse neonatal outcome (i.e., LGA) was correlated with increased/excessive fetal movements, particularly when the gestational historic period was more than 37 weeks. An improved understanding into the cause of excessive fetal activities and outcomes would let future studies to investigate whether reduced adverse neonatal outcomes can exist accomplished past encouraging women to report excessive fetal activities followed by appropriate intervention. This would also provide translational value to modifiable adventure factors for improving perinatal and neonatal outcomes.
Availability of information and materials
The datasets generated and/or analysed during the current study are not publicly available due to results are obtained from identifying images and/or other clinical details of participants that may compromise anonymity, but are available from the corresponding author on reasonable request.
Abbreviations
- APGAR:
-
Appearance, pulse, grimace, action, and respiration
- CI:
-
Confidence interval
- LGA:
-
Big for gestational age
- NST:
-
Non-stress test
- OR:
-
Odds ratio
- STARS:
-
Situation, task, actions, and results
References
-
Lawn JE, Blencowe H, Waiswa P, Amouzou A, Mathers C, Hogan D, et al. Stillbirths: rates, gamble factors, and dispatch towards 2030. Lancet. 2016;387:587–603.
-
Stacey T, Thompson J, Mitchell EA, Ekeroma AJ, Zuccollo JM, McCowan LM. The Auckland stillbirth study, a case–control study exploring modifiable risk factors for third trimester stillbirth: methods and rationale. Aust N Z J Obstet Gynaecol. 2011;51:three–8.
-
Platts J, Mitchell EA, Stacey T, Martin BL, Roberts D, McCowan L, et al. The Midland and due north of England stillbirth study (MiNESS). BMC Pregnancy Childbirth. 2014;14:171.
-
Gordon A, Raynes-Greenow C, Bond D, Morris J, Rawlinson W, Jeffery H. Sleep position, fetal growth restriction, and belatedly-pregnancy stillbirth: the Sydney stillbirth report. Obstet Gynecol. 2015;125:347–55.
-
Heazell AP, Frøen J. Methods of fetal motility counting and the detection of fetal compromise. J Obstet Gynaecol. 2008;28:147–54.
-
Unterscheider J, Horgan R, O'Donoghue K, Greene R. Reduced fetal movements. Obstet Gynaecol. 2009;11:245–51.
-
McCarthy CM, Meaney S, O'Donoghue K. Perinatal outcomes of reduced fetal movements: a cohort study. BMC Pregnancy Childbirth. 2016;16:169.
-
Poojari VG, Kumar SS, Vasudeva A. Obstetric and neonatal outcome among women presenting with reduced fetal movements in third trimester. Int J Reprod Contracept Obstet Gynecol. 2017;seven:88–93.
-
Warrander LK, Heazell AE. Identifying placental dysfunction in women with reduced fetal movements can exist used to predict patients at increased take a chance of pregnancy complications. Med Hypotheses. 2011;76:17–20.
-
Olagbuji B, Igbarumah S, Akintayo A, Olofinbiyi B, Aduloju P, Alao O. Maternal understanding of fetal motion in third trimester: a means for fetal monitoring and reducing stillbirth. Niger J Clin Pract. 2014;17:489–94.
-
Warland J, O'Brien LM, Heazell AE, Mitchell EA. An international cyberspace survey of the experiences of 1,714 mothers with a belatedly stillbirth: the STARS cohort study. BMC Pregnancy Childbirth. 2015;15:172.
-
Linde A, Pettersson M, Rådestad I. Women's experiences of fetal movements before the confirmation of fetal death—contractions misinterpreted as fetal movement. Nativity. 2015;42:189–94.
-
Luoto R, Kinnunen TI, Aittasalo 1000, Kolu P, Raitanen J, Ojala K, et al. Primary prevention of gestational diabetes mellitus and big-for-gestational-age newborns past lifestyle counseling: a cluster-randomized controlled trial. PLoS Med. 2011;8:e1001036.
-
Surkan PJ, Hsieh C-C, Johansson AL, Dickman Prisoner of war, Cnattingius S. Reasons for increasing trends in large for gestational age births. Obstet Gynecol. 2004;104:720–six.
-
Kim SY, Sharma AJ, Sappenfield W, Wilson HG, Salihu HM. Association of maternal body mass index, excessive weight gain, and gestational diabetes mellitus with large-for-gestational-age births. Obstet Gynecol. 2014;123:737.
-
Leipold H, Worda C, Gruber CJ, Kautzky-Willer A, Husslein PW, Bancher-Todesca D. Large-for-gestational-historic period newborns in women with insulin-treated gestational diabetes under strict metabolic control. Wien Klin Wochenschr. 2005;117:521–five.
-
Dubil EA, Magann EF. Amniotic fluid as a vital sign for fetal wellbeing. Aust J Ultrasound Med. 2013;sixteen:62–70.
-
Krishna U, Bhalerao S. Placental insufficiency and fetal growth brake. J Obstet Gynecol Bharat. 2011;61:505–11.
-
Stacey T, Thompson J, Mitchell EA, Ekeroma A, Zuccollo J, McCowan LM. Maternal perception of fetal action and late stillbirth adventure: findings from the Auckland stillbirth report. Nativity. 2011;38:311–6.
-
Heazell AE, Warland J, Stacey T, Coomarasamy C, Budd J, Mitchell EA, et al. Stillbirth is associated with perceived alterations in fetal activity–findings from an international instance control written report. BMC Pregnancy hildbirth. 2017;17:369.
-
Greenish-top guideline No R. 57: reduced fetal movements. London: Royal College of Obstetricians and Gynaecologists; 2011. p. 2016.
-
Hofmeyr GJ, Novikova N. Direction of reported decreased fetal movements for improving pregnancy outcomes. Cochrane Database Syst Rev. 2012;4:CD009148.
Acknowledgements
Not applicable.
Funding
This work was funded by Shanghai Municipal Didactics Commission-Gaoyuan Nursing Grant Support (project: Hlgy1816ky). The funding body played no function in the design of the study and drove, assay, and estimation of data and in writing the manuscript.
Author data
Affiliations
Contributions
Information collection and analysis: CH, WH; Study designed and manuscript writing: YF. All authors read and approved the final version of the manuscript.
Corresponding author
Ideals declarations
Ideals approval and consent to participate
All participants have given informed and written consent. This written report was approved past the ethics committee of Shanghai Sixth People's Hospital, Shanghai Jiao Tong Academy, and followed the ethical guidelines laid downwards in the 1975 Declaration of Helsinki.
Consent for publication
Not Applicable.
Competing interests
The authors declare that they take no competing interests.
Additional data
Publisher'south Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open up Access This article is distributed under the terms of the Artistic Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted employ, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/ane.0/) applies to the data made available in this commodity, unless otherwise stated.
Reprints and Permissions
Near this commodity
Cite this article
Huang, C., Han, W. & Fan, Y. Correlation study between increased fetal motility during the third trimester and neonatal result. BMC Pregnancy Childbirth nineteen, 467 (2019). https://doi.org/ten.1186/s12884-019-2637-4
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1186/s12884-019-2637-iv
Keywords
- Fetal movements
- Neonatal outcome
- Large for gestational age
- Correlational report
Source: https://bmcpregnancychildbirth.biomedcentral.com/articles/10.1186/s12884-019-2637-4
0 Response to "Baby Size and Fetal Movement at 37 Weeks"
Post a Comment